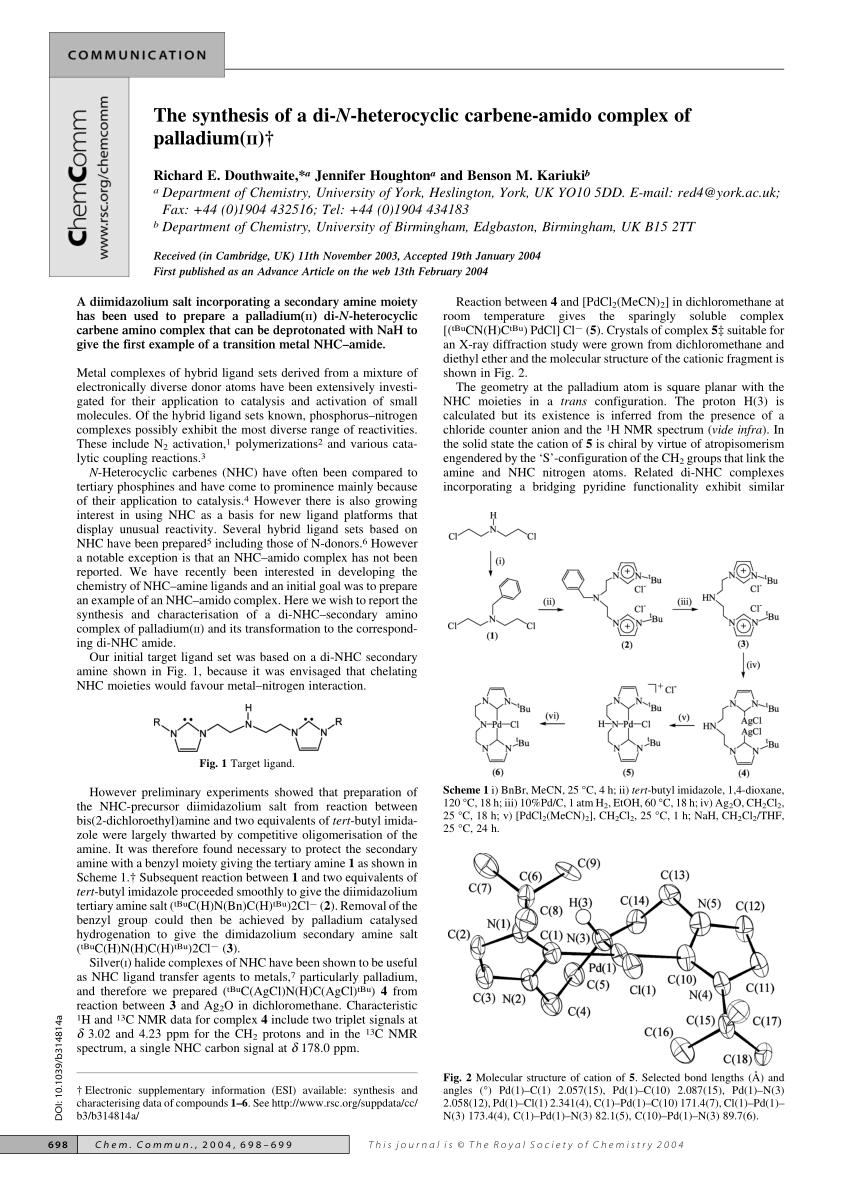
Palladium–Carbene Complexes Supported by Polyphenylene Matrices as Catalysts for THE Suzuki, Heck and cyanation reactions, INEOS OPEN, 2018, 1 (2), 103–111
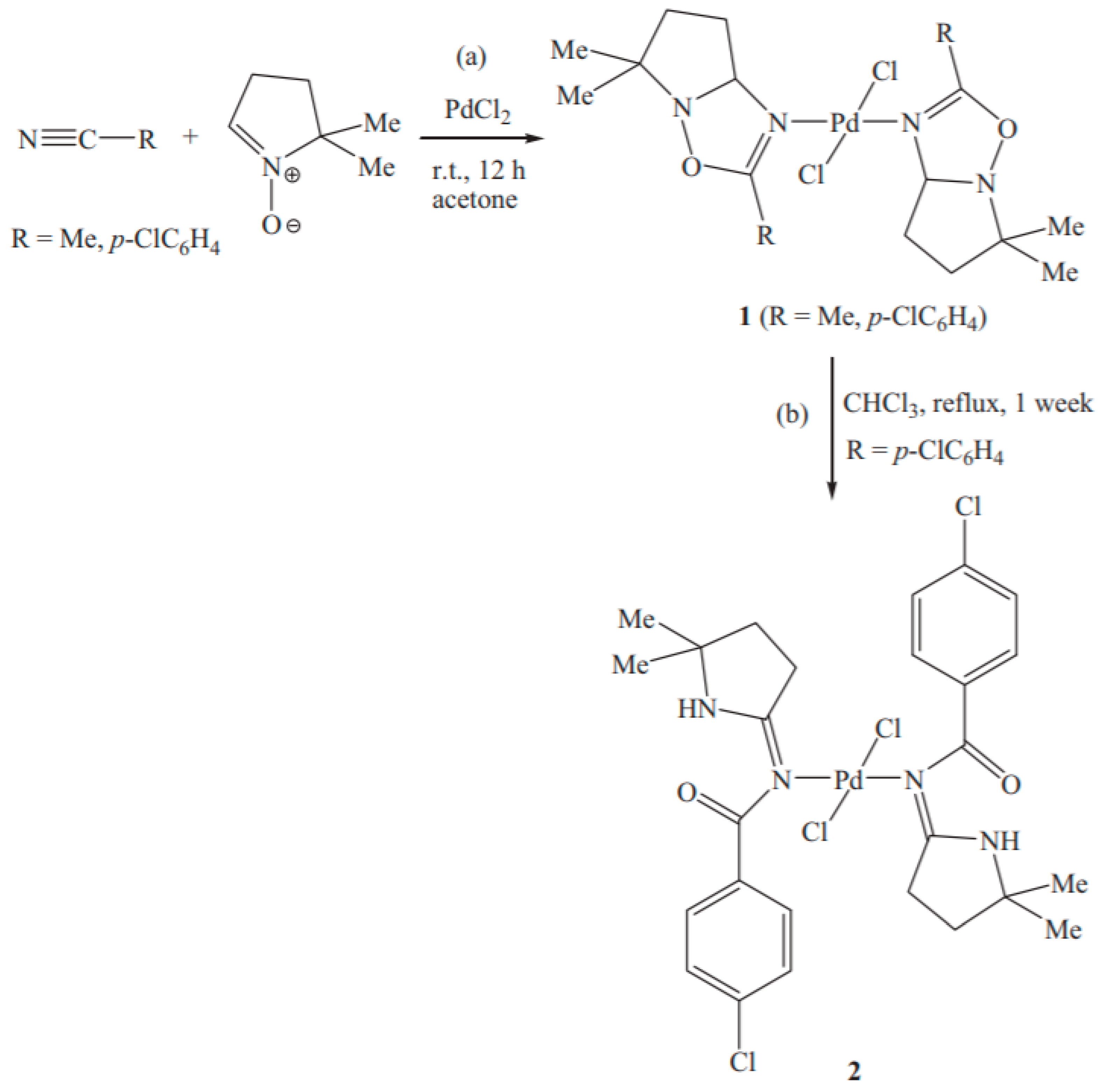
Catalysts | Free Full-Text | Recent Developments in the Immobilization of Palladium Complexes on Renewable Polysaccharides for Suzuki–Miyaura Cross-Coupling of Halobenzenes and Phenylboronic Acids | HTML

Palladium Complexes Bearing Mesoionic Carbene Ligands: Applications in α‐Arylation, α‐Methylation and Suzuki–Miyaura Coupling Reactions - Maity - 2016 - European Journal of Inorganic Chemistry - Wiley Online Library
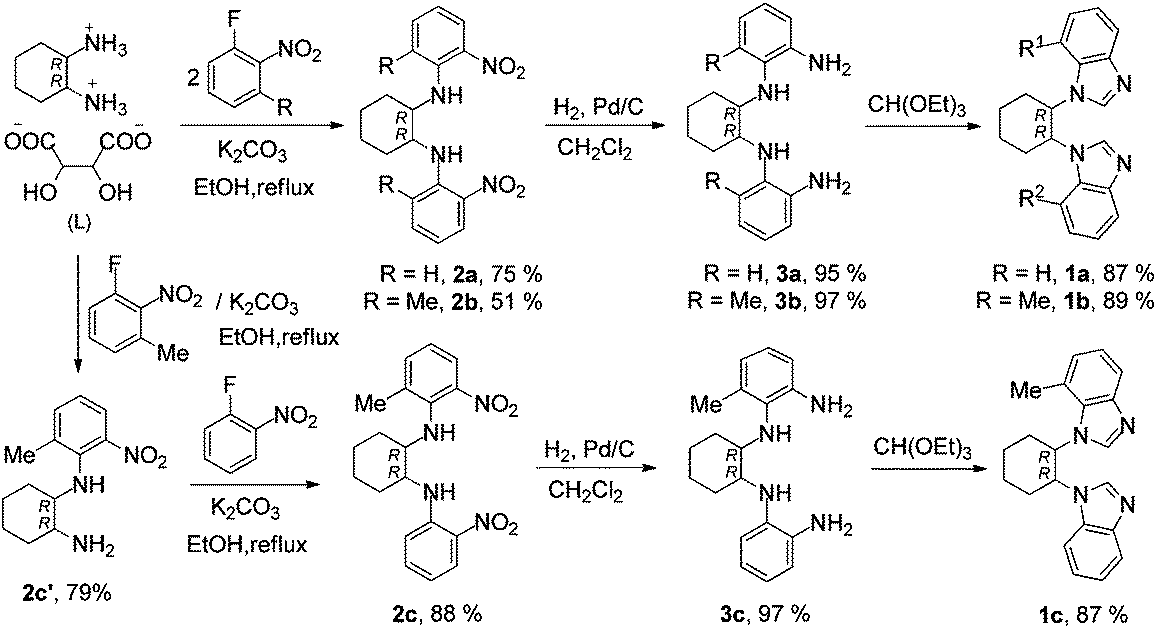
Chiral linker-bridged bis-N-heterocyclic carbenes: design, synthesis, palladium complexes, and catalytic properties - Dalton Transactions (RSC Publishing)
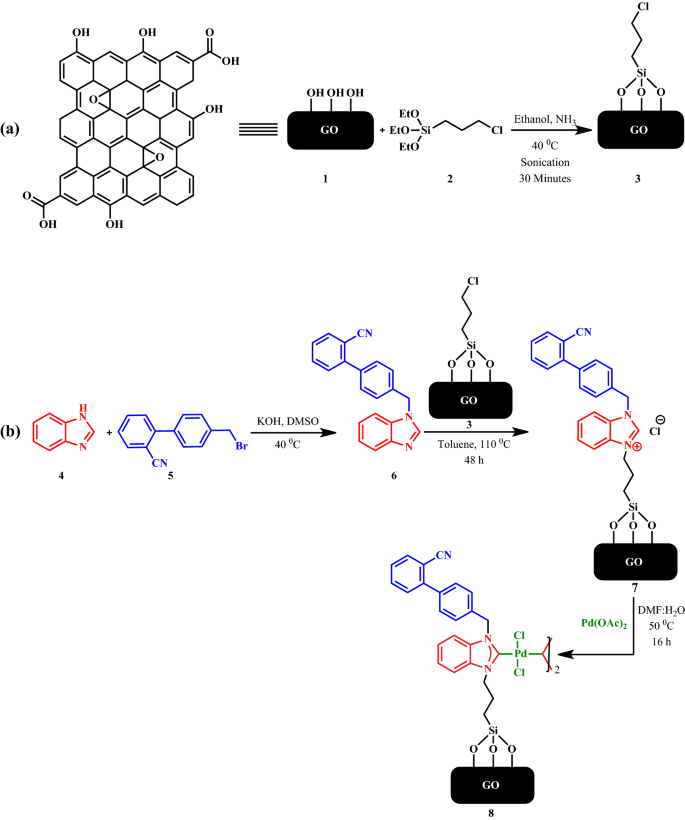
Immobilized N -Heterocyclic Carbene-Palladium(II) Complex on Graphene Oxide as Efficient and Recyclable Catalyst for Suzuki–Miyaura Cross-Coupling and Reduction of Nitroarenes | SpringerLink
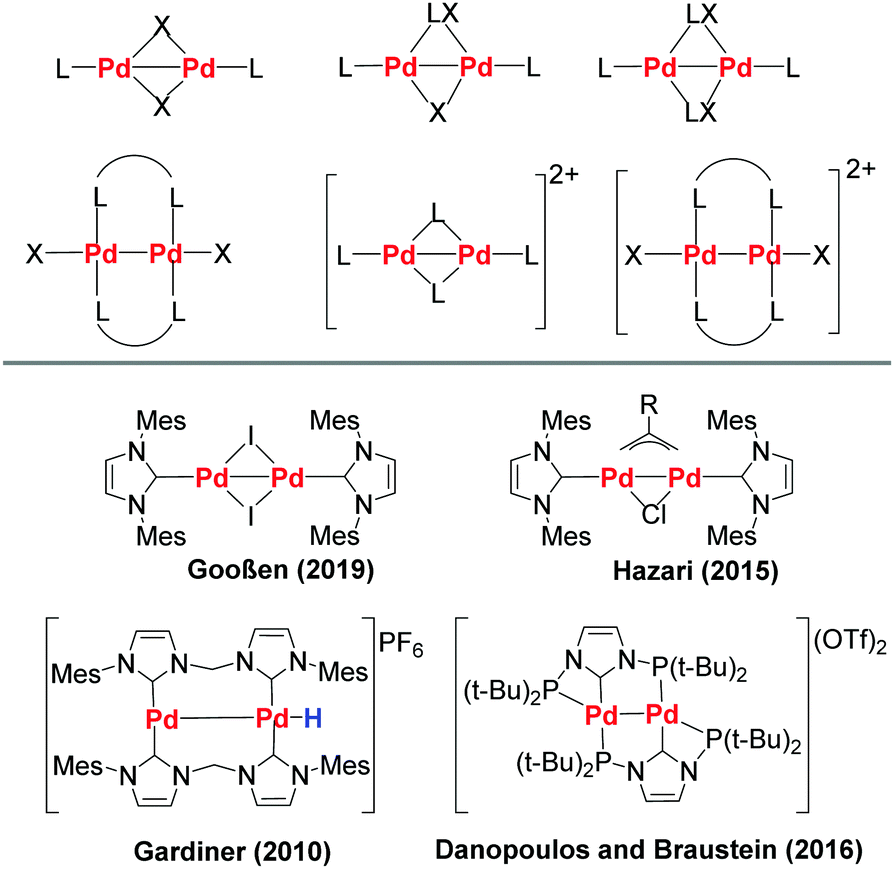
The anticancer activity of an air-stable Pd(i)-NHC (NHC = N-heterocyclic carbene) dimer - Chemical Communications (RSC Publishing)

Reactive Palladium Carbenes: Migratory Insertion and Other Carbene–Hydrocarbyl Coupling Reactions on Well‐Defined Systems - Albéniz - 2018 - European Journal of Inorganic Chemistry - Wiley Online Library

The first used butylene linked bis(N-heterocyclic carbene)-palladium-PEPPSI complexes in the direct arylation of furan and pyrrole - ScienceDirect
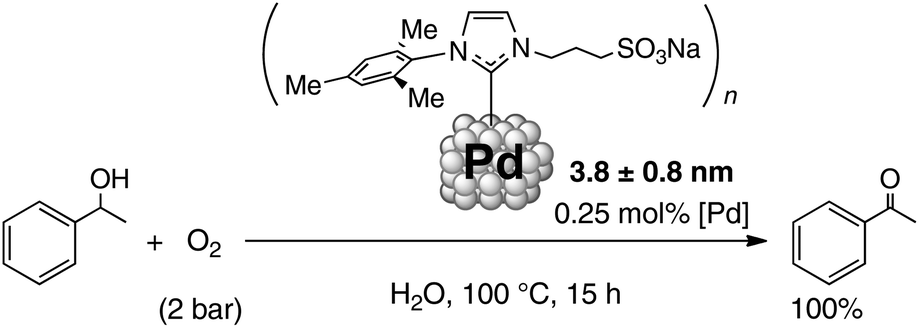
Synthesis of water-soluble palladium( ii ) complexes with N-heterocyclic carbene chelate ligands and their use in the aerobic oxidation of 1-phenyleth ... - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT00643H

Palladium–Carbene Complexes Supported by Polyphenylene Matrices as Catalysts for THE Suzuki, Heck and cyanation reactions, INEOS OPEN, 2018, 1 (2), 103–111

The anticancer activity of an air-stable Pd(i)-NHC (NHC = N-heterocyclic carbene) dimer - Chemical Communications (RSC Publishing)

Palladium–Carbene Complexes Supported by Polyphenylene Matrices as Catalysts for THE Suzuki, Heck and cyanation reactions, INEOS OPEN, 2018, 1 (2), 103–111

PDF) Palladium(II) N -heterocyclic carbene complexes: Synthesis, structures and cytotoxicity potential studies against breast cancer cell line
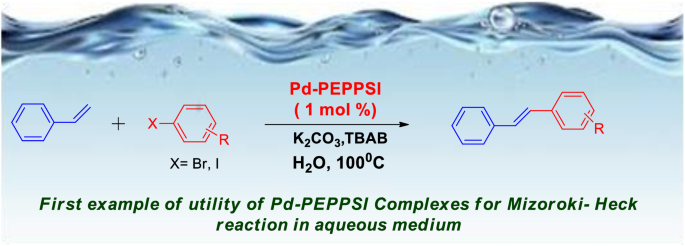
A new PEPPSI type N-heterocyclic carbene palladium(II) complex and its efficiency as a catalyst for Mizoroki-Heck cross-coupling reactions in water | SpringerLink

Reactive Palladium Carbenes: Migratory Insertion and Other Carbene–Hydrocarbyl Coupling Reactions on Well‐Defined Systems - Albéniz - 2018 - European Journal of Inorganic Chemistry - Wiley Online Library

Palladium–Carbene Complexes Supported by Polyphenylene Matrices as Catalysts for THE Suzuki, Heck and cyanation reactions, INEOS OPEN, 2018, 1 (2), 103–111

Palladium–Carbene Complexes Supported by Polyphenylene Matrices as Catalysts for THE Suzuki, Heck and cyanation reactions, INEOS OPEN, 2018, 1 (2), 103–111
Palladium complexes of N-heterocyclic carbenes displaying an unsymmetrical N-alkylfluorenyl/N′-aryl substitution pattern and their behaviour in Suzuki–Miyaura cross coupling - Dalton Transactions (RSC Publishing)
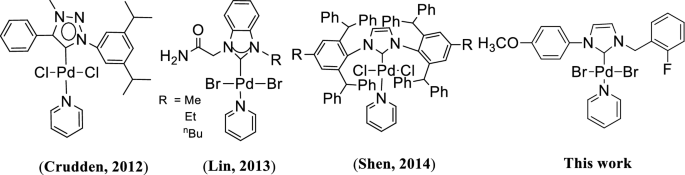
A new PEPPSI type N-heterocyclic carbene palladium(II) complex and its efficiency as a catalyst for Mizoroki-Heck cross-coupling reactions in water | SpringerLink
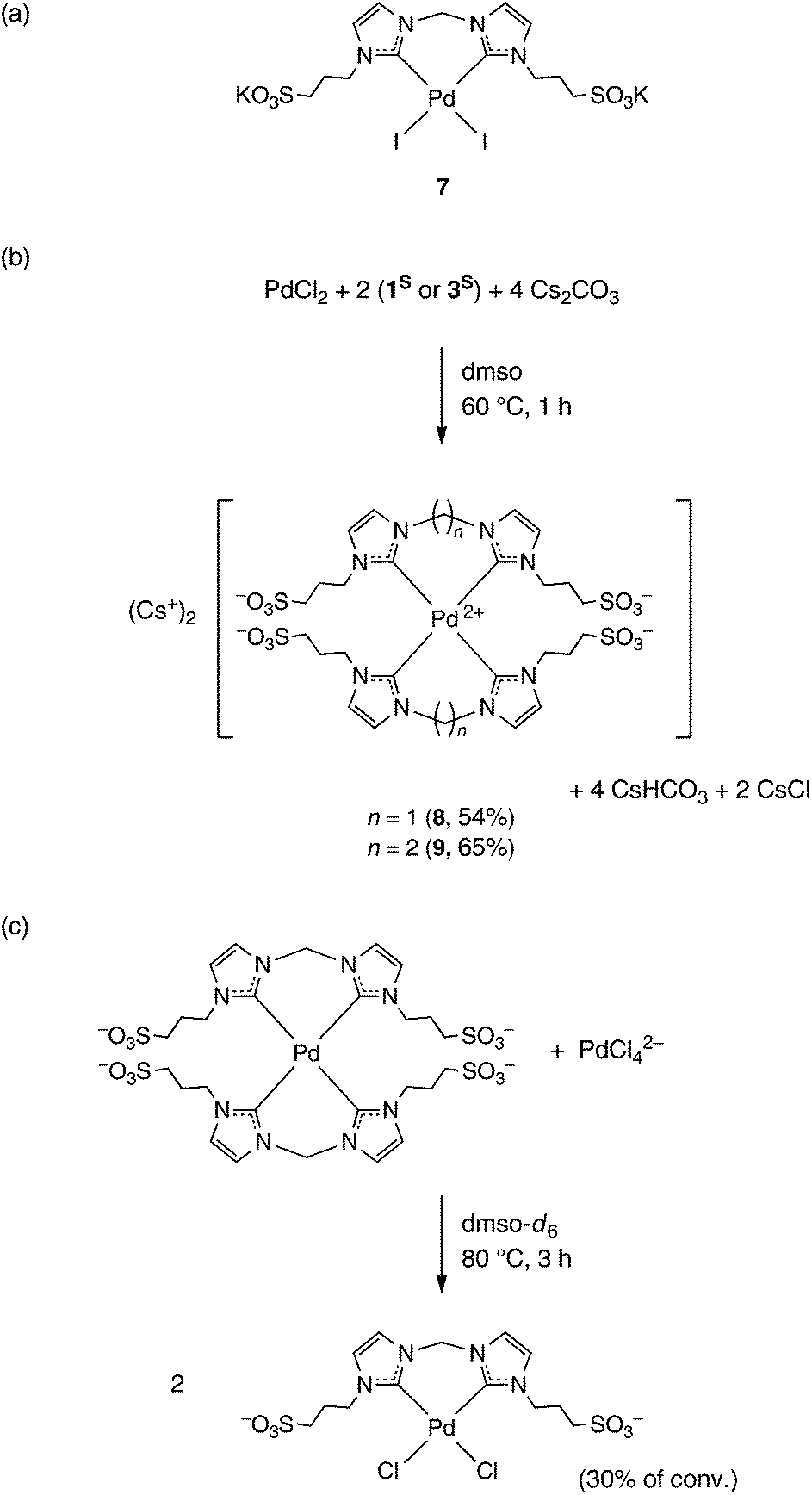
Synthesis of water-soluble palladium( ii ) complexes with N-heterocyclic carbene chelate ligands and their use in the aerobic oxidation of 1-phenyleth ... - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT00643H

Shape-Controllable Formation of Poly-imidazolium Salts for Stable Palladium N -Heterocyclic Carbene Polymers | Scientific Reports
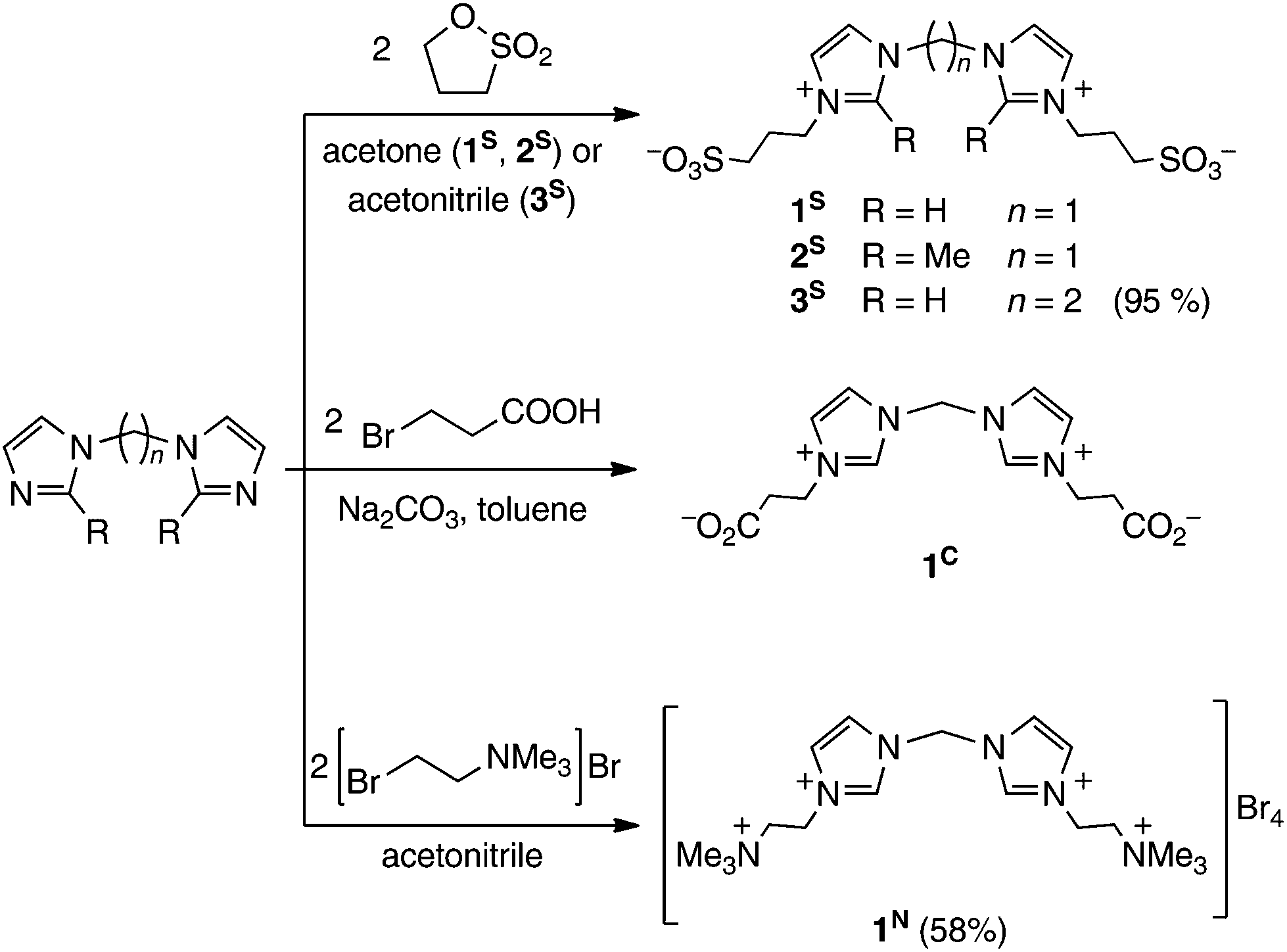
Synthesis of water-soluble palladium( ii ) complexes with N-heterocyclic carbene chelate ligands and their use in the aerobic oxidation of 1-phenyleth ... - Dalton Transactions (RSC Publishing) DOI:10.1039/C7DT00643H
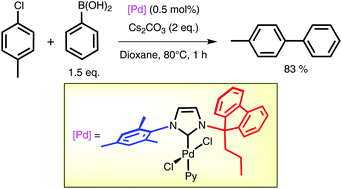
Palladium complexes of N-heterocyclic carbenes displaying an unsymmetrical N-alkylfluorenyl/N′-aryl substitution pattern and their behaviour in Suzuki–Miyaura cross coupling - Dalton Transactions (RSC Publishing)