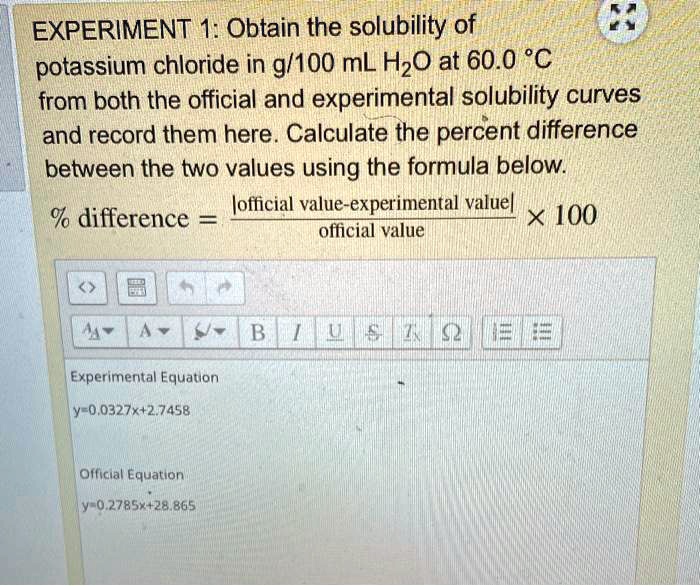
SOLVED: (E1) (E2) (E3) (E4) Fi2 k"r12 E=kCg;; 5 = f E"dA Lnc E = k f %; (C1) C2 C3 C4) C5 C6) (E5) (EG) (E7) Vi = k94 V=kCL V =

Strategies To Control Product Characteristics in Simultaneous Crystallization of NaCl and KCl from Aqueous Solution: Seeding with KCl | Crystal Growth & Design

Amazon.com: BulkSupplements.com Potassium Chloride Powder - Potassium Supplement for Muscle Support & Electrolytes - Gluten Free, Soy Free, and No Filler Powder, Potassium Salt (500 Grams - 1.1 lbs) : Health & Household

Distinction between Human Cytochrome P450 (CYP) Isoforms and Identification of New Phosphorylation Sites by Mass Spectrometry | Journal of Proteome Research
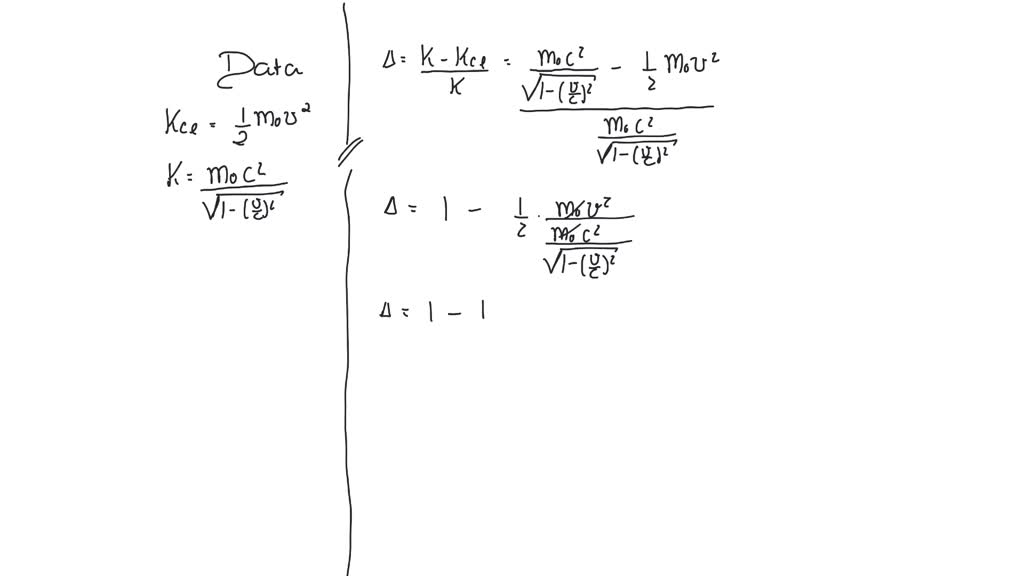
SOLVED: What is the percent difference between the classical kinetic energy, Kcl=12m0v2, and the correct relativistic kinetic energy, K=m0c2/1−v2/c2−−−−−−−√−m0c2, at a speed of 0.11 c? Express your answer using two significant figures.

Question Video: Determining the Mass of the Potassium Chloride Analyte When 2g of Silver Chloride Precipitates | Nagwa

Atomic insights into flotation separation of KCl and NaCl from a new viewpoint of hydration layer: A molecular dynamic study - ScienceDirect
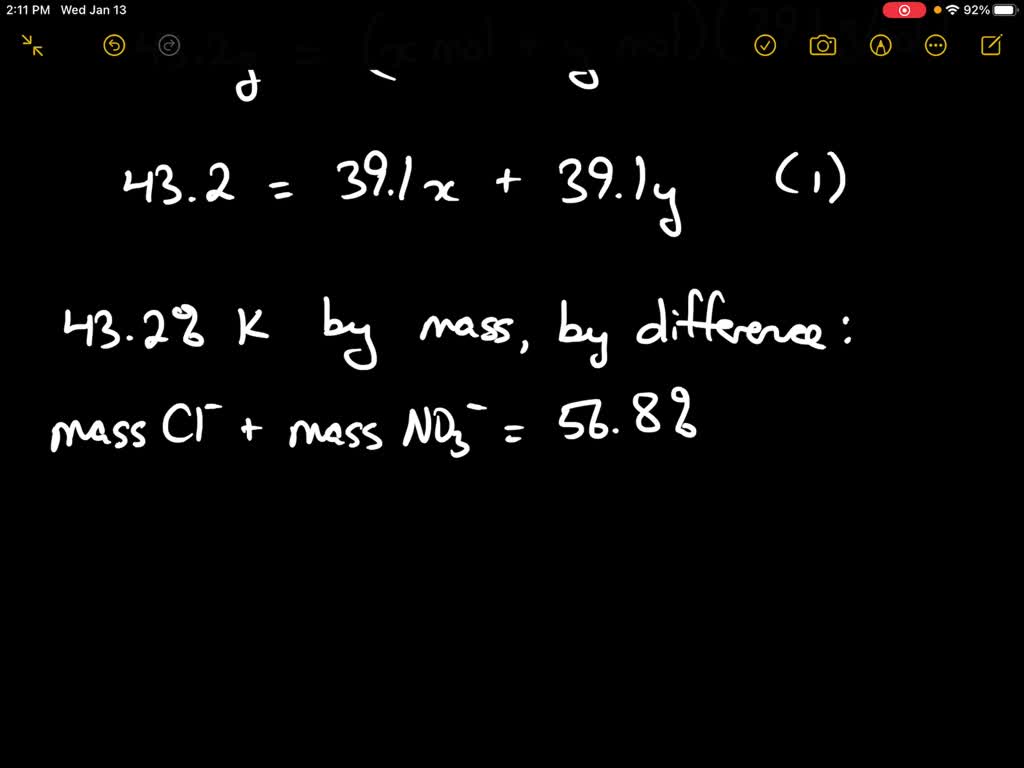
SOLVED: El peso molecular del KCl (Cloruro de potasio)es: * 74,54 g/mol 35,45 g/mol 73, 54 g/mol 46,45 g/mol