
Calculate the half life of a first order reaction from their rate constants given below:(a) 200 s^-1 ; (b) 2 min^-1 ; (c) 4 year^-1 .
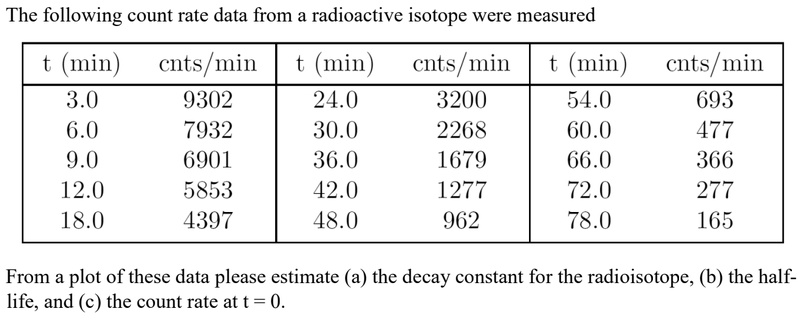
SOLVED: The following count rate data from a radioactive isotope were measured (min) 3.0 6.0 9.0 12.0 18.0 cnts /min 9302 7932 6901 5853 4397 t (min) cnts /min 24.0 3200 30.0

For the elementary reaction 2A → C ,the concentration of A after 30 minutes was found to be 0.01 mole/lit. If the rate constant of the reaction is 2.5 × 10^-2 lit

Half life of a first order reaction is 2.1xx10^(12)s. Calculate the rate constant of the reactio... - YouTube
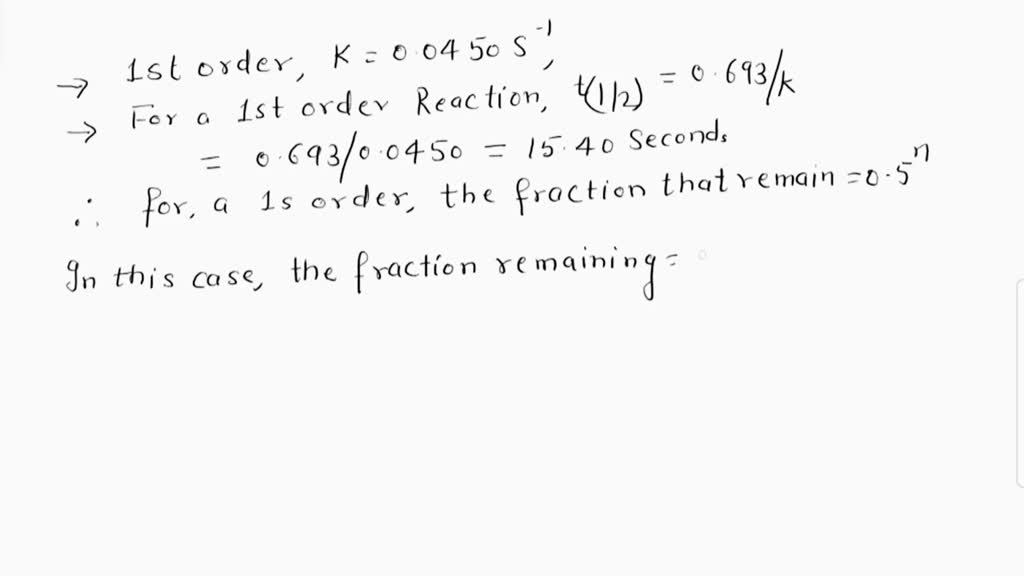
SOLVED: If a reaction is first order with a rate constant of 0.0450 s⁻¹, how much time is required for 65% of the initial quantity of reactant to be consumed?

Chapter 9, Chemical Kinetics Video Solutions, Numerical problems in Physical Chemistry for IIT-JEE | Numerade
A first order reaction completes 50% at the end of 50 minutes. What is the value of rate constant in sec^-1? How many times will the reaction be complete at 87.5%? - Quora

Calculate the half life of a first order reaction from their rate constants given below:(a) 200 s^-1 ; (b) 2 min^-1 ; (c) 4 year^-1 .

The energy of activation and specific rate constant for a first order reaction at 25^∘ C are 100kJ/mole and 3.46 × 10^-5sec^-1 respectively. Determine the temperature at which half - life of

The unit of first order rate constant when concentration is measured in terms of pressure and time - YouTube

The reaction is given below, the rate constant for disappearance of A is 7.48 × 10^-3sec^-1 . The time required for the total pressure in a system containing A at an initial
![a) For a reaction ABrarrP , the rate law is given by, r=k[A]^(1//2)[B]^(2). What is the order of this reaction? (b) A first order reaction is found to have a rate constant a) For a reaction ABrarrP , the rate law is given by, r=k[A]^(1//2)[B]^(2). What is the order of this reaction? (b) A first order reaction is found to have a rate constant](https://d10lpgp6xz60nq.cloudfront.net/ss/web/885563.jpg)
a) For a reaction ABrarrP , the rate law is given by, r=k[A]^(1//2)[B]^(2). What is the order of this reaction? (b) A first order reaction is found to have a rate constant

In the following gaseous phase first order reaction A(g) → 2 B(g) + C(g) initial pressure was found to be 400 mm of Hg and it changed to 1000 mm of Hg

SOLVED: The potassium isotope K-40 undergoes beta decay with a half-life of 1.83*10^9 years. Find the number of beta decays that occur per second in 1.0g of pure K-40.

The reaction `A(g)toB(g)+2C(g)` is a first order reaction with rate constant `2.772xx10^(-3)sec^(-1) - YouTube





